
Momentum Microsystems is a proven industry leader in global OEM integration services with more than 20 years of experience partnering with medical OEMs, ISVs, manufacturers and logistics service providers.
We deliver turnkey computing solutions, end-to-end product integration, global supply chain and support services across the globe.
Our processes enable our customers to stay focused on enhancing their intellectual property, building competitive advantage and driving business outcomes while leaving the operational execution to our highly skilled engineering and operations teams.

Momentum is proud to be an HP Amplify Impact Catalyst 3-Star Partner. Learn more >>
For over two decades Momentum has built long-lasting partner relationships with leading computing and software vendors.


Mindesk is a 3D design developer enabling architects, designers, and engineers to build CAD projects in multi-user VR environments.


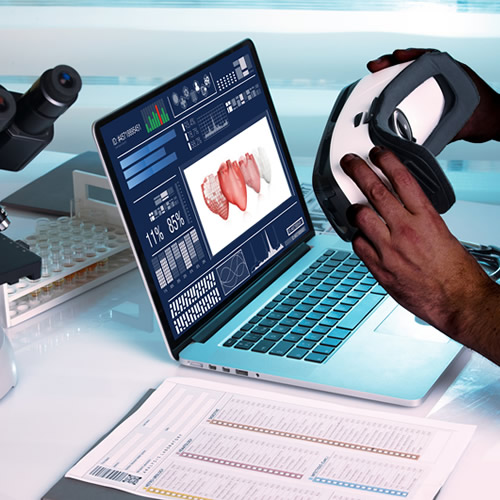
Our highly skilled engineering team collaborates with you throughout the product design process. We provide development and market insights which improve the design, quality and manufacturability of your product. The result is industry-leading turnkey integrated solutions delivered in a time- and cost-effective manner.
Time to market is now more critical than ever. Momentum is NPI made easy!
“Momentum Micro allows us to focus on our intellectual property and ensure that our solution will meet all global regulatory compliance”
– OEM Biotech Customer